Scheibye-Knudsen Group
In the Scheibye-Knudsen Group we try to understand the cellular and organismal consequences of DNA damage with the aim of developing interventions.

The growing proportion of the elderly population represents an increasing socioeconomic burden, not least because of age-associated diseases. It is therefore increasingly pertinent to find interventions that may lead to interventions for age-associated diseases such as Alzheimer's, Parkinson's and cardiovascular diseases. Although the cause of aging is currently unknown accumulation of damage to our genome, the DNA, may be an contributing factor.
In the Scheibye-Knudsen lab we try to understand the cellular and organismal consequences of DNA damage with the aim of developing interventions. We have discovered that DNA damage leads to changes in certain metabolites and that replenishment of these molecules may alter the rate of aging in model organisms. These findings suggest that normal aging and age-associated diseases may be malleable to similar interventions. The hope is to develop interventions that will allow everyone to live healthier, happier and more productive lives.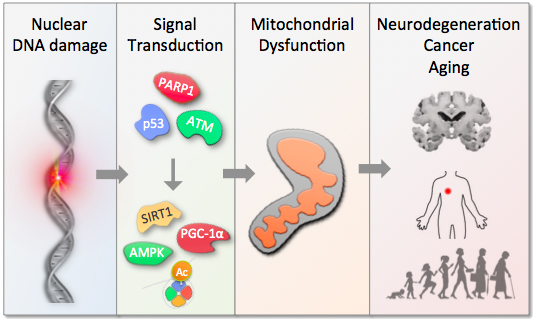
"To discover interventions leading to healthier, happier and more productive lives"
Aging represents the largest risk factor for most chronic diseases. Notably, the proportion of elderly is increasing across the globe and thus most societies face a significant socio-economic burden of an aging population. Although the cause of aging is unknown recent work suggests that signaling from damaged DNA may be an important factor driving organismal aging.
Importantly, we and others have found that interventions in DNA damage signaling may stall the aging process leading to increased health and longevity. The overall goal of the lab is to further understand the aging process and to develop interventions that may lead to healthier aging.
The goal is pursued through a variety of ways including in silico analyses, in vitro biochemistry and molecular biology as well as in vivo work on mouse models.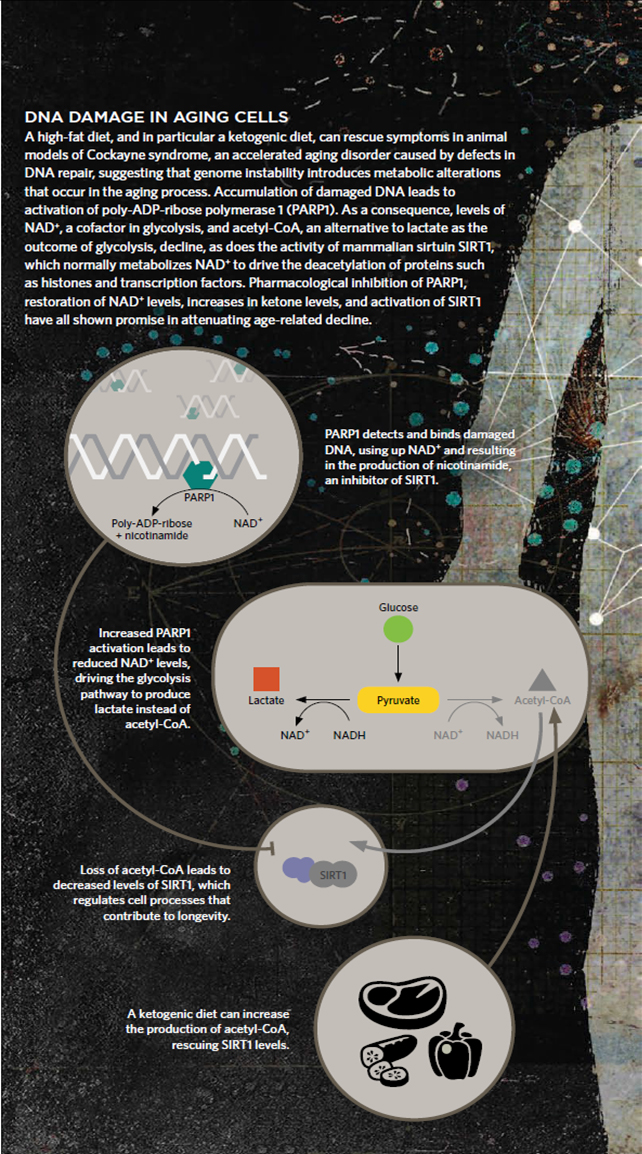
 Group Leader:
Group Leader:
Morten Scheibye-Knudsen
Associate Professor
Email: mscheibye@sund.ku.dk
Phone: +45 42 50 70 60
LinkedIn: Morten’s profile
Twitter: @ScheibyeKnudsen
Members of the Scheibye-Knudsen group
| Name | Title | Phone | |
|---|---|---|---|
| Garik Mkrtchyan | Assistant Professor | +4535333040 | |
| Michael Ben Ezra | Postdoc | +4535335290 | |
| Morten Scheibye-Knudsen | Associate Professor | +4535337096 |
